The Monash Milk Team is part of the Boyd group at Monash Institute of Pharmaceutical Sciences – click here to go to our broader group’s website.
Publications


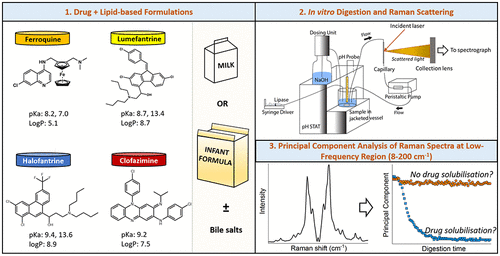
Salim M., Fraser-Miller S.J., Rziņš K.B., Sutton J.J., Ramirez G., Clulow A.J., Hawley A., Beilles S., Gordon K.C., Boyd B.J., Low-frequency raman scattering spectroscopy as an accessible approach to understand drug solubilization in milk-based formulations during digestion, Molecular Pharmaceutics 2020, 17, 885 - 899
Li T., Clulow A.J., Nowell C.J., Hawley A., Cipolla D., Rades T., Boyd B.J., Controlling the size and shape of liposomal ciprofloxacin nanocrystals by varying the lipid bilayer composition and drug to lipid ratio, Journal of Colloid and Interface Science 2019, 555, 361 - 372
Salim M., Ramirez G., Clulow A.J., Zhang Y., Ristroph K.D., Feng J., McManus S.A., Hawley A., Prud'Homme R.K., Boyd B.J., Solid-State Behavior and Solubilization of Flash Nanoprecipitated Clofazimine Particles during the Dispersion and Digestion of Milk-Based Formulations, Molecular Pharmaceutics 2019, 16, 2755 - 2765
Salim M., Fraser-Miller S.J., Sutton J.J., Berzinš K., Hawley A., Clulow A.J., Beilles S., Gordon K.C., Boyd B.J., Application of Low-Frequency Raman Scattering Spectroscopy to Probe in Situ Drug Solubilization in Milk during Digestion, Journal of Physical Chemistry Letters 2019, 10, 2258 - 2263
Salim M., Khan J., Ramirez G., Murshed M., Clulow A.J., Hawley A., Ramachandruni H., Beilles S., Boyd B.J., Impact of Ferroquine on the Solubilization of Artefenomel (OZ439) during in Vitro Lipolysis in Milk and Implications for Oral Combination Therapy for Malaria, Molecular Pharmaceutics 2019, 16, 1658 - 1668
Binte Abu Bakar S.Y., Salim M., Clulow A.J., Hawley A., Boyd B.J., Revisiting dispersible milk-drug tablets as a solid lipid formulation in the context of digestion, International Journal of Pharmaceutics 2019, 554, 179 - 189
Pham A., Gavin P.D., Libinaki R., Ramirez G., Khan J.T., Boyd B.J., Differential effects of TPM, a phosphorylated tocopherol mixture, and other tocopherol derivatives as excipients for enhancing the solubilization of co-enzyme Q10 as a lipophilic drug during digestion of lipid-based formulations, Current Drug Delivery 2019, 16, 628 - 636
Boyd B.J., Salim M., Clulow A.J., Ramirez G., Pham A.C., Hawley A., The impact of digestion is essential to the understanding of milk as a drug delivery system for poorly water soluble drugs, Journal of Controlled Release 2018, 292, 13 - 17
Salim M., Khan J., Ramirez G., Clulow A.J., Hawley A., Ramachandruni H., Boyd B.J., Interactions of Artefenomel (OZ439) with Milk during Digestion: Insights into Digestion-Driven Solubilization and Polymorphic Transformations, Molecular Pharmaceutics 2018, 15, 3535 - 3544
Clulow A.J., Salim M., Hawley A., Gilbert E.P., Boyd B.J., The Curious Case of the OZ439 Mesylate Salt: An Amphiphilic Antimalarial Drug with Diverse Solution and Solid State Structures, Molecular Pharmaceutics 2018, 15, 2027 - 2035
Clulow A.J., Salim M., Hawley A., Boyd B.J., A closer look at the behaviour of milk lipids during digestion, Chemistry and Physics of Lipids 2018, 211, 107 - 116
Pham A.C., Gavin P., Libinaki R., Ramirez G., Boyd B.J., A new lipid excipient, phosphorylated tocopherol mixture, TPM enhances the solubilisation and oral bioavailability of poorly water soluble CoQ10 in a lipid formulation, Journal of Controlled Release 2017, 268, 400 - 406
Clulow A.J., Parrow A., Hawley A., Khan J., Pham A.C., Larsson P., Bergström C.A.S., Boyd B.J., Characterization of Solubilizing Nanoaggregates Present in Different Versions of Simulated Intestinal Fluid, Journal of Physical Chemistry B 2017, 121, 10869 - 10881